Unveiling the Mysteries of Radon: A Closer Look at the Noble Gas
Radon:
Nestled inconspicuously in the noble gases group of the periodic table, Radon stands out as a unique and intriguing element. Symbolized by Rn and boasting an atomic number of 86, Radon is an odorless, colorless, and tasteless gas that has captivated the interest of scientists and researchers alike. In this blog, we will explore the various facets of Radon, from its chemical and physical properties to its compounds, reactions, occurrence, production, and practical applications.
Chemical and Physical Properties:
Latin name: Radon
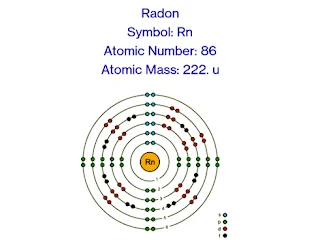
Symbol: Rn
Atomic Mass: 86
Atomic Number: 222 u
Electron configuration short: [Xe] 4f14 5d10 6s2 6p6
Electron configuration
in full form: 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 4f14 5s2 5p6 5d10 6s2 6p6
Valence Electron: 8
Valency: 0
As a noble gas, Radon exhibits characteristics typical of its group. It has a Latin name derived from the word 'radium,' its precursor element. With an atomic mass of approximately 222 atomic mass units, Radon is a monatomic gas with a single atom in its elemental form. it holds a full complement of electrons in its outer shell, rendering it stable and unreactive.
Despite being inert, Radon is radioactive, and its isotopes emit alpha particles during decay. This property contributes to its potential health risks, particularly when it accumulates in confined spaces.
Radon Compounds:
In its elemental state, Radon rarely forms compounds due to its inert nature. However, under extreme conditions, researchers have managed to synthesize a few compounds, such as radon fluorides. These compounds are highly unstable and serve mainly as subjects of scientific investigation.
Chemical Reactions:
Due to its noble gas status, Radon is generally unreactive and does not readily engage in chemical reactions with other elements. However, in laboratory settings, scientists have induced reactions with highly reactive substances to form temporary compounds.
Occurrence and Production:
Radon is not naturally occurring in significant quantities but is a decay product of heavier elements such as uranium and thorium. It forms through the decay of radium, a process that contributes to its presence in the Earth's crust, soil, and rock. Radon can seep into buildings, particularly in regions with high concentrations of uranium and thorium.
The production of Radon in a controlled environment involves extracting it from uranium-containing minerals or through artificial means. The latter process often involves bombarding bismuth-209 with high-energy neutrons in nuclear reactors.
Uses and Facts:
While Radon is primarily known for its potential health risks due to its radioactivity, it also finds applications in various fields. Radon testing is crucial in assessing indoor air quality, particularly in areas where the gas may accumulate in buildings. Additionally, Radon-222 has been utilized in certain medical treatments for its alpha-emitting properties.
Conclusion:
Radon, with its unique combination of properties, remains an enigmatic element in the periodic table. From its inert nature to its radioactivity, Radon poses both challenges and opportunities for scientific exploration. As we delve deeper into the mysteries of this noble gas, our understanding continues to evolve, shedding light on its significance in the realms of health, science, and technology.
Also Read:









0 Comments